
How medical device manufacturing can be brought back to the US

How medical device manufacturing can be brought back to the US
Cardiosense appoints John Martin as CMO, enhancing AI-driven cardiac care and advancing regulatory efforts for innovative hemodynamic monitoring solutions.

SetPoint Medical secures $140 million to launch innovative neuroimmune modulation therapy for rheumatoid arthritis, advancing treatment for autoimmune diseases.

Discover how Orpyx's innovative sensory insoles advance diabetes foot care, enhancing health outcomes and reducing costs through real-time monitoring and support.
Apreo Health pioneers emphysema treatment with innovative device trials, aiming to transform care for millions suffering from COPD.
Ventric Health advances heart failure detection with the Vivio System, enabling accurate diagnoses in primary care for improved heart health outcomes.

TAO Digital enhances its medtech expertise by acquiring DigitalSoftMinds, boosting capabilities in medical devices and AI-powered solutions for regulated industries.

Yale begins enrolling patients in PRIME study using next-generation Swoop system

Rowan University and NJEDA launch a Strategic Innovation Center to boost medical device investments and accelerate medtech innovation in South Jersey.

Cardiosense's CardioTag gains FDA clearance, advancing cardiac monitoring with wearable technology for personalized, noninvasive heart health insights.
CMS establishes a national payment for AI ECG analysis, paving the way for enhanced cardiovascular diagnostics and early patient intervention.
RenovoRx launches the PanTheR study to evaluate the RenovoCath device's long-term safety and effectiveness in treating solid tumors, advancing cancer care.

Edwards Lifesciences reports robust Q2 growth, raises 2025 guidance, driven by strong TAVR and TMTT sales, enhancing its market leadership.

MeMed BV Flex could revolutionize infection diagnosis, enabling rapid differentiation between bacterial and viral infections in just 15 minutes with a fingerstick test.

FDA's AI tool, Elsa, faces scrutiny for producing unreliable data, raising concerns about its rapid implementation and impact on regulatory accuracy.

GE HealthCare leads in FDA AI-enabled medical device authorizations, showcasing innovation in imaging and diagnostics to enhance patient care and streamline workflows.

Digital asthma program shows sustained benefits in landmark clinical trial

New 154,000-square-foot Class A facility aims to address the growing demand for purpose-built life sciences manufacturing infrastructure.

Independent practices say EHRs lead the way — but telehealth, patient portals and analytics tools are driving measurable results, especially in mental health care.
Proximie and HistoSonics collaborate to enhance non-invasive liver tumor treatments, improving patient outcomes and surgical efficiency through advanced technology integration.

Caranx Medical's AI software advances TAVI procedures, enhancing precision and accessibility for heart valve surgeries, improving patient outcomes.

iFIT and Samsung Health unite to deliver personalized fitness experiences through Galaxy devices, enhancing wellness with AI-driven insights and interactive content.
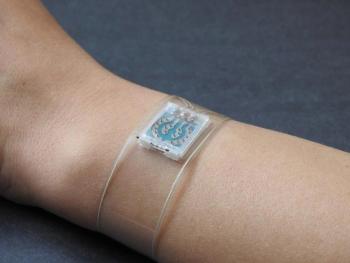
A flexible new wristband developed by engineers at the University of California San Diego could improve how people with diabetes manage their health by continuously monitoring glucose and key cardiovascular signals in real time.
Bruin Biometrics CEO Martin Burns discusses the "formulas" that guided the company through the challenges of bringing a device to market.