Article
UPDATE: Focus on practice
Special report: A boost for patient safety practices; formularies; self-referral; malpractice awards; physicians marching; abortion
UPDATE
Focus on Practice
By Joan R. Rose
Formularies for Medicaid patients are okay, this court says
States are free to establish preferred drug lists and prior authorization programs for Medicaid beneficiaries, a federal court has ruled. In the suit, filed last year, the Pharmaceutical Research and Manufacturers of America asked the court to invalidate Michigan's Pharmaceutical Best Practices Initiative program and to prohibit HHS Secretary Tommy Thompson from approving similar programs. To get their drugs on the Michigan formulary, pharmaceutical manufacturers must grant rebates beyond those already required by the Medicaid program.
More than a dozen states have announced or already implemented programs similar to Michigan's. Now Michigan plans to partner with Vermont and South Carolina to implement the nation's first multistate purchasing arrangement for pharmaceuticals paid for under their respective Medicaid programs.
Stark sets the stage for Act III
Rep. Pete Stark (D-CA) once again has doctors' investments in his sights. He and Rep. Jerry Kleczka (D-WI) want to build on previous laws governing physician self-referral (Stark I and Stark II) with the Hospital Investment Act of 2003, which aims to close a loophole that currently allows physicians to refer patients to some health facilities from which they benefit financially.
Under the bill, physicians would not be allowed to refer patients to a hospital in which they've invested unless the general public had the same opportunity to invest in the venture. Failure to comply would result in financial penalties; in addition, both the physicians and the hospitals would be barred from participation in the Medicare program.
As currently written, the measure would affect not only planned and future investments in specialty hospitals, but also any transaction that occurred in the past few years, according to Charles Oppenheim, a health care attorney with Foley & Lardner. Oppenheim notes that even hospitals that have had physician investors for many years would be affectedif and when these hospitals seek new physician investors, or an existing investor attempts to sell his interest to another physician investor.
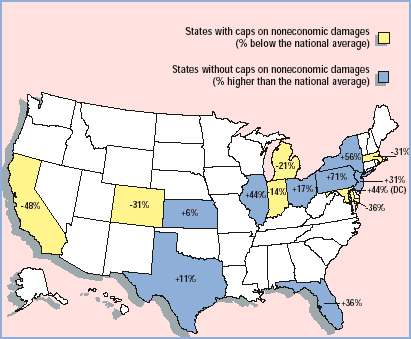
Do caps on damages reduce malpractice costs?
Critics have claimed that caps on noneconomic damages will do nothing to cut the cost of insuring physicians against medical malpractice, but a study by Milliman USA suggests otherwise.
The Seattle-based actuarial consulting firm analyzed medical malpractice claims closed from late 1990 to early 2001 in the 14 largest states and the District of Columbia. Analysts found that the cost of malpractice losses (based on statistics reported to the National Practitioner Data Bank) were below average in states that had laws limiting noneconomic damages, while states without these reforms had losses above the national average.
Although there are other differences between the statesincluding the size and application of the capsthe study "very clearly shows that caps on noneconomic damages are highly correlated to medical malpractice costs," says Richard S. Biondi, author of the study.
Liability premiums send physicians marching
Doctors continue to take to the streets and even close their offices to patients over the professional liability insurance crisis. With the federal tort reform bill stalled in Congress, doctors are seeking relief from state legislatures with increasing fervor.
The hottest action is in New Jersey. A work action that shut offices around the state for several days culminated on Feb. 4 in a rally at the state capital in Trenton and led to a compromise billwith a $300,000 cap on noneconomic damages and a limited catastrophic fundthat passed the state Senate. When the state Assembly dawdled, some 8,000 to 10,000 doctors marched back to Trenton on May 15one of the largest job actions ever organized by US physiciansand promised a more-drastic work stoppage if the Assembly didn't pass tort reform.
Also in early May, several thousand Pennsylvania physicians refused to see patients, while surgeons and ob/gyns in Jacksonville, FL, closed offices.
Doctors in Connecticut, New York, and Chicago have rallied to call the attention of their legislators to the crisis, and surgeons in West Virginia staged a hospital walkout in January to protest soaring malpractice premiums.
The abortion debate takes on a circus atmosphere
Minnesota lawmakers tacked an abortion bill on to one that deregulates circuses, and the governor signed the measure. The amendment requires women to receive certain information (such as the gestational age of the fetus) and then wait 24 hours before undergoing the procedure.
NARAL Pro-Choice America's Minnesota affiliate lobbied heavily to defeat the measure, and painted the state's pro-choice governor as having caved in to "the extreme agenda of the religious right."
SPECIAL REPORT A boost for patient safety practices
The National Quality Forum, a private-sector, Washington, DC-based organization created in 1999 to develop and implement a national strategy for health care quality measurement and reporting, last month released a report that identified 30 evidence-based "safe practices" for health care settings. Here are the systems and procedures most applicable to office-based physician practices:
Create a health care culture of safety.
For designated high-risk, elective surgical procedures and other specified care, patients should be clearly informed of the likely reduced risk of an adverse outcome at treatment facilities that have demonstrated superior outcomes and should be referred to such facilities in accordance with the patient's stated preference.
Specify an explicit protocol to be used to ensure an adequate level of nursing care based on the institution's usual patient mix and the experience and training of its nursing staff.
All patients in general intensive care units (both adult and pediatric) should be managed by physicians having specific training and certification in critical care medicine.
Pharmacists should actively participate in the medication-use process, including, at a minimum, being available for consultation with prescribers on medication ordering, interpretation and review of medication orders, preparation of medications, dispensing of medications, and administration and monitoring of medications.
Verbal orders should be recorded whenever possible and immediately read back to the prescriberi.e., a health care provider receiving a verbal order should read or repeat back the information that the prescriber conveys in order to verify the accuracy of what was heard.
Use only standardized abbreviations and dose designations.
Patient care summaries or similar records should not be prepared from memory.
Ensure that care information, especially changes in orders and new diagnostic information, is transmitted in a timely and clearly understandable form to all of the patient's current health care providers/professionals who need that information to provide care.
Ask each patient or legal surrogate to recount what he or she has been told during the informed consent discussion.
Ensure that written documentation of the patient's preference for life-sustaining treatment is prominently displayed in his or her chart.
Implement a computerized prescriber order entry system.
Implement standardized protocols to prevent the occurrence of wrong-site procedures or wrong-patient procedures.
Evaluate each patient undergoing elective surgery for risk of an acute ischemic cardiac event during surgery, and provide prophylactic treatment of high-risk patients with beta blockers.
Utilize dedicated antithrombotic (anticoagulation) services that facilitate coordinated care management.
Utilize validated protocols to evaluate patients who are at risk for contrast media-induced renal failure, and utilize a clinically appropriate method for reducing risk of renal injury based on the patient's kidney function evaluation.
Decontaminate hands with either a hygienic hand rub or by washing with a disinfectant soap prior to and after direct contact with the patient or objects immediately around the patient.
Vaccinate health care workers against influenza to protect both them and patients from influenza.
Keep workspaces where medications are prepared clean, orderly, well lit, and free of clutter, distraction, and noise.
Standardize the methods for labeling, packaging, and storing medications.
Identify all "high alert" drugs (e.g., intravenous adrenergic agonists and antagonists, chemotherapy agents, anticoagulants and antithrombotics, concentrated parenteral electrolytes, general anesthetics, neuromuscular blockers, insulin and oral hypoglycemics, narcotics and opiates).
Dispense medication in unit-dose or, when appropriate, unit-of-use form, whenever possible.
For more information about the National Quality Forum, visit www.qualityforum.org.
Our Web Poll
Joan Rose. UPDATE: Focus on practice.
Medical Economics
Jun. 6, 2003;80:10.





